Your searched for: quick gliatm astrocytes human ipsc derived astrocytes f 21 yr donor healthy control as sev cw70067
-
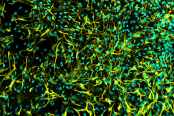
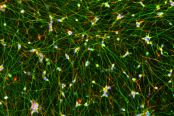
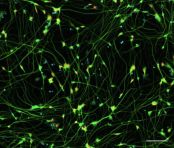
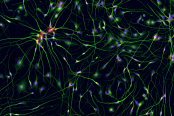
AS-SeV-HCCryopreserved human iPSC-derived astrocytes from Healthy Control donor
-
AS-SeV-CW50065Cryopreserved human iPSC-derived astrocytes from Healthy Control patient
-
AS-SeV-ADCryopreserved human iPSC-derived astrocytes from Alzheimer's Disease patient
-
AS-SeV-EPCryopreserved human iPSC-derived astrocytes from Epilepsy patient
-
AS-SeV-AUCryopreserved human iPSC-derived astrocytes from Autism Spectrum Disorder patient
-
AS-SeVThis kit differentiates human pluripotent stem cells into astrocytes in 28 days using Sendai virus.
-
EX-SeV-HCCryopreserved human iPSC-derived excitatory neurons from Healthy Control donor
-
MT-SeV-HCCryopreserved human iPSC-derived motor neurons from Healthy Control donor
-
CH-SeV-HCCryopreserved human iPSC-derived cholinergic neurons from Healthy Control donor
-
DA-mRNA-HCCryopreserved human iPSC-derived Dopaminergic neurons from Healthy Control donor